"Jim Kress" <nos### [at] kressworks com> wrote in message
news:3d3a5242@news.povray.org...
> This is the electrostatic potential of a new, never before synthesized
> 'triple decker' organometallic 'sandwich compound', superimposed on its
> electron density. The synthesis and structure of the compound was just
> recently published in Angew. Chem. Int. Ed. 2002, 41, No. 9.
I can't read the article (or can you reach it via internet?), so I ask you
directly. Is it one central atom (or ion) or are there two of them? And
what is it? Magnesium or a heavier kind?
> I calculated the electron density and molecular electrostatic potential
> using my normal set of Quantum Chemical software tools.
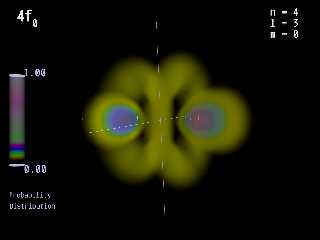
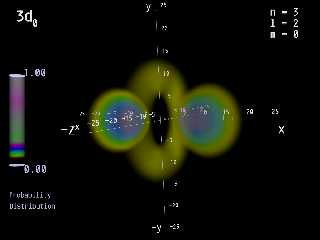
I am interested in this software. I have made pictures of the orbitals of
hydrogen for my students at school (see attachments) with formula's I
digged out of a very old quantum mechanic book that I stumbled over
in a second hand bookstore years ago. If it is possible to render
pictures of molecules with Molecular Orbitals with that software,
maybe I can persuade my school to buy it. Computer graphics make
students to take interest in subjects they otherwise ignore.
Further on you talk about electrostatic potential. Is it correct that you
mean with that the energy potential of a atom caused by the net charge
of the nucleus and his electrons?
If so, is it possible to calculate this for bounded atoms as well, using
Molecular Orbitals?
Regards,
Jaap Frank com> wrote in message
news:3d3a5242@news.povray.org...
> This is the electrostatic potential of a new, never before synthesized
> 'triple decker' organometallic 'sandwich compound', superimposed on its
> electron density. The synthesis and structure of the compound was just
> recently published in Angew. Chem. Int. Ed. 2002, 41, No. 9.
I can't read the article (or can you reach it via internet?), so I ask you
directly. Is it one central atom (or ion) or are there two of them? And
what is it? Magnesium or a heavier kind?
> I calculated the electron density and molecular electrostatic potential
> using my normal set of Quantum Chemical software tools.
I am interested in this software. I have made pictures of the orbitals of
hydrogen for my students at school (see attachments) with formula's I
digged out of a very old quantum mechanic book that I stumbled over
in a second hand bookstore years ago. If it is possible to render
pictures of molecules with Molecular Orbitals with that software,
maybe I can persuade my school to buy it. Computer graphics make
students to take interest in subjects they otherwise ignore.
Further on you talk about electrostatic potential. Is it correct that you
mean with that the energy potential of a atom caused by the net charge
of the nucleus and his electrons?
If so, is it possible to calculate this for bounded atoms as well, using
Molecular Orbitals?
Regards,
Jaap Frank
Post a reply to this message
Attachments:
Download 'H_PROB_4F0_HIGH.PNG' (136 KB)
Download 'H_PROB_3D0_HIGH.PNG' (138 KB)
Preview of image 'H_PROB_4F0_HIGH.PNG'
Preview of image 'H_PROB_3D0_HIGH.PNG'
|




![]()